Just dug an old chair out
53 posts
• Page 1 of 2 • 1, 2
Just dug an old chair out
from the back of the garage that was abandoned 2 years back. It has 2 old odyssey batteries in it, that read 10V and 8.8V each.
They have been uncharged for 2 years, and are now way beyond 100% discharged.
I connected in parallel, and am charging at 40A and 14.7V. With Shirley POWER SUPPLY/charger. Fan runs at same speed as when idle. Stone cold. Batteries didn't want to charge, stupid low current. So I set it to 30V for a while. And it rose to the full 40A and 18V. So now reset back to correct 14.70V.
After 30 mins at 14.7V its charging at a miserable 6A. So very sulfated. So I increased to 15.6V. Now charging at 11A. 5.5A each battery. And falling... They are unwell.
So in an attempt to desulfate as much as possible I have set it to 17v Max, and a CC of 8A so 4A per battery, and will continue this until the batts have received 140% capacity. So 68Ah, each PLUS 27Ah, so almost 200Ah total.
They have been uncharged for 2 years, and are now way beyond 100% discharged.
I connected in parallel, and am charging at 40A and 14.7V. With Shirley POWER SUPPLY/charger. Fan runs at same speed as when idle. Stone cold. Batteries didn't want to charge, stupid low current. So I set it to 30V for a while. And it rose to the full 40A and 18V. So now reset back to correct 14.70V.
After 30 mins at 14.7V its charging at a miserable 6A. So very sulfated. So I increased to 15.6V. Now charging at 11A. 5.5A each battery. And falling... They are unwell.
So in an attempt to desulfate as much as possible I have set it to 17v Max, and a CC of 8A so 4A per battery, and will continue this until the batts have received 140% capacity. So 68Ah, each PLUS 27Ah, so almost 200Ah total.
-

Burgerman - Site Admin
- Posts: 65240
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Just dug an old chair out
3 hour on, and its beginning to take a charge. So set instead to 40A and 15V. Currently taking 15A. And so far a supposed 24Ah...
-

Burgerman - Site Admin
- Posts: 65240
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Just dug an old chair out
Electricity and time are worth more than new batteries...
-

martin007 - Posts: 3320
- Joined: 03 Jun 2015, 23:55
- Location: Spain
Re: Just dug an old chair out
Yes. But just to find out...
-

Burgerman - Site Admin
- Posts: 65240
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Just dug an old chair out
GRANVILLE BAT-AID BATTERY AID TABLET
5 new from £3.60
Breaks down deposits on battery plates
Extends the life of the battery
Restores battery power
Suitable for lead acid batteries
But no good for sealed batteries unless you can get the tops of the cells off
5 new from £3.60
Breaks down deposits on battery plates
Extends the life of the battery
Restores battery power
Suitable for lead acid batteries
But no good for sealed batteries unless you can get the tops of the cells off
-

Mechniki - Posts: 272
- Joined: 22 Oct 2013, 21:59
- Location: Dorking, Surrey
Re: Just dug an old chair out
Its no good for any batteries. Even wet acid ones they were intended for. They are snake oil.
-

Burgerman - Site Admin
- Posts: 65240
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Just dug an old chair out
so you trying to cook off
-

Mechniki - Posts: 272
- Joined: 22 Oct 2013, 21:59
- Location: Dorking, Surrey
Re: Just dug an old chair out
No.
HERE is how it works...
A lead battery has positive and negative plates both made from lead. Usually nasty contaminated reclaimed lead, but Odyssey batter=ies use 99.99+ pure virgin lead. So when they are abandoned or left discharged they will sulfate just the same as all lead batteries. But unlike those which also deteriorate due to masses of unwanted reactions from the acid and impurity metals, or added metals like calcium or zinc (added to strengthen the nasty reclaimed lead plates).
In addition to get a lot of surface area from these lead plates they are not flat they are a grid like this:

Each plate then has FINELY ground up mix of electrolyte and lead "pasted" literally into the holes. This is because all those lead particles have MUCH more surface area than a flat lead plate. So now they look like this showing some un-pasted grids,(plates) and some pasted ones :

These will then have seperators made of Plastic mesh in wet acid (electrolyte), and gel batteries.
And glass fibre tissue in an AGM battery soaked in electrolyte.
Many of these lead grid/lead (lead oxide) paste plates are fitted into each cell with a separator in between. So the acid, or gel, can be in between each plate and in full contact with it.
In an AGM battery like the Odyssey.
In all 3 batteries the electrolyte is a mix of water, and Sulfuric acid.
In the case of a gel battery some silica jel crystals are also added to make the electrolyte into a very thick paste. Its like butter from the fridge but almost clear.
****************
Now, when the fully charged battery is discharged, it does the following.
Lead and lead dioxide, the active materials on the battery's plates, react with sulfuric acid in the electrolyte to form lead sulfate as the battery becomes DISCHARGED. That lead sulfate is important!!!
The lead sulfate first forms in a finely divided, amorphous state, and easily reverts to lead, lead dioxide and sulfuric acid when the battery is recharged. Provided it is FULLY recharged. Which takes 12 to 16 hours minimum.
Sulfation then is the result of discharging. The electrolyte gets weaker as you discharge, because it is removed from the electrolyte leaving mostly water. Which doesn't conduct well... As long as you recharge RAPIDLY, and very COMPLETELY then this lead sulfate is no problem, and is returned to the acid where it belongs. The problem is this.
If you leave a battery for any length of time at anything less than 100% fully charged, this means some lead sulfate coating remains on the plate and this turns to larger crystals over time. This is now called permanent sulfation. Because these larger crystals do not conduct electricity at all. So no amount of charging, pulsing, battery re-conditioners, desulfators, or pills, can help. The more sulfation remaining, that cannot be returned to the acid, on the worse the battery gets. In the end, it goes into the bin.
The problem is that as well as sulfation, the plates become distorted. And the active lead and lead oxide material on the plates becomes used up, and pushed apart as it expands with the growth of the crystals. And so this, along with all the impurities and unwanted reactions mean that even if you were able to desulfate the battery it still ruined in other ways too.
But the pure lead Odyssey (and the same with Optima) means that the lead can be made much thinner and still retain strength. This also means no unwanted reactions as all the impurities are absent. And that sulfation does less damage to the active materials as it thinner. And near the surface. And the AGM construction means that the plates are packed close together and under pressure. So cant distort like traditional plates. And the same with the spiral plates on the Optima. Super thin and self supporting. So when a battery is allowed to discharge deeply, for too long, the plates cant distort. And theres no unwanted reactions. So its WORTH trying to add extra voltage, and charging for a LONG time in an attempt to turn all the sulfates that can be, back into acid. Theres far more chance of success. As less damage happens.
The best way to attemp this, on a tired battery or one thats been abused like these, is to use a small CONSTANT CURRENT of about an amp per battery. For a long time, and allow enough volts to begin with to keep this 1A working. Allow a Max voltage initially of up to 16 or 17V. As soon as the current reaches 1A the voltage will fall. So on a power supply you would set 1A max. 17V. And leave it for around 1.4x its capacity in Ah at this fixed current. And then add another 10 hours if its voltage starts to drop below 13V when off charge.. Then test.
HERE is how it works...
A lead battery has positive and negative plates both made from lead. Usually nasty contaminated reclaimed lead, but Odyssey batter=ies use 99.99+ pure virgin lead. So when they are abandoned or left discharged they will sulfate just the same as all lead batteries. But unlike those which also deteriorate due to masses of unwanted reactions from the acid and impurity metals, or added metals like calcium or zinc (added to strengthen the nasty reclaimed lead plates).
In addition to get a lot of surface area from these lead plates they are not flat they are a grid like this:

Each plate then has FINELY ground up mix of electrolyte and lead "pasted" literally into the holes. This is because all those lead particles have MUCH more surface area than a flat lead plate. So now they look like this showing some un-pasted grids,(plates) and some pasted ones :

These will then have seperators made of Plastic mesh in wet acid (electrolyte), and gel batteries.
And glass fibre tissue in an AGM battery soaked in electrolyte.
Many of these lead grid/lead (lead oxide) paste plates are fitted into each cell with a separator in between. So the acid, or gel, can be in between each plate and in full contact with it.
In an AGM battery like the Odyssey.
In all 3 batteries the electrolyte is a mix of water, and Sulfuric acid.
In the case of a gel battery some silica jel crystals are also added to make the electrolyte into a very thick paste. Its like butter from the fridge but almost clear.
****************
Now, when the fully charged battery is discharged, it does the following.
Lead and lead dioxide, the active materials on the battery's plates, react with sulfuric acid in the electrolyte to form lead sulfate as the battery becomes DISCHARGED. That lead sulfate is important!!!
The lead sulfate first forms in a finely divided, amorphous state, and easily reverts to lead, lead dioxide and sulfuric acid when the battery is recharged. Provided it is FULLY recharged. Which takes 12 to 16 hours minimum.
Sulfation then is the result of discharging. The electrolyte gets weaker as you discharge, because it is removed from the electrolyte leaving mostly water. Which doesn't conduct well... As long as you recharge RAPIDLY, and very COMPLETELY then this lead sulfate is no problem, and is returned to the acid where it belongs. The problem is this.
If you leave a battery for any length of time at anything less than 100% fully charged, this means some lead sulfate coating remains on the plate and this turns to larger crystals over time. This is now called permanent sulfation. Because these larger crystals do not conduct electricity at all. So no amount of charging, pulsing, battery re-conditioners, desulfators, or pills, can help. The more sulfation remaining, that cannot be returned to the acid, on the worse the battery gets. In the end, it goes into the bin.
The problem is that as well as sulfation, the plates become distorted. And the active lead and lead oxide material on the plates becomes used up, and pushed apart as it expands with the growth of the crystals. And so this, along with all the impurities and unwanted reactions mean that even if you were able to desulfate the battery it still ruined in other ways too.
But the pure lead Odyssey (and the same with Optima) means that the lead can be made much thinner and still retain strength. This also means no unwanted reactions as all the impurities are absent. And that sulfation does less damage to the active materials as it thinner. And near the surface. And the AGM construction means that the plates are packed close together and under pressure. So cant distort like traditional plates. And the same with the spiral plates on the Optima. Super thin and self supporting. So when a battery is allowed to discharge deeply, for too long, the plates cant distort. And theres no unwanted reactions. So its WORTH trying to add extra voltage, and charging for a LONG time in an attempt to turn all the sulfates that can be, back into acid. Theres far more chance of success. As less damage happens.
The best way to attemp this, on a tired battery or one thats been abused like these, is to use a small CONSTANT CURRENT of about an amp per battery. For a long time, and allow enough volts to begin with to keep this 1A working. Allow a Max voltage initially of up to 16 or 17V. As soon as the current reaches 1A the voltage will fall. So on a power supply you would set 1A max. 17V. And leave it for around 1.4x its capacity in Ah at this fixed current. And then add another 10 hours if its voltage starts to drop below 13V when off charge.. Then test.
-

Burgerman - Site Admin
- Posts: 65240
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Just dug an old chair out
One battery is all but charged, down to .3A at 14.7v.
The other still 3A at the same voltage. But it is still falling slowly, 22 hours later.
Will discharge about 10Ah (if it has 10Ah!) and recharge again. Chair can be driven...
The other still 3A at the same voltage. But it is still falling slowly, 22 hours later.
Will discharge about 10Ah (if it has 10Ah!) and recharge again. Chair can be driven...
-

Burgerman - Site Admin
- Posts: 65240
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Just dug an old chair out
Battery was not great. It didn't want to drop down to the 1000thC termination point, and didn't have much capacity on test.
So I charged again, at 14.70v till current stopped falling. CV for 12 hours. Then set it to 15.6V, and to 1A and left it for another 12 hours. Nothing to lose. Remember that this is 2 batts in parallel.
Obviously it was deeply sulfated. So slightly more volts, constant 1A current to desulfate it as far as possible. Because current is limited to .5A per battery, it can cope with the gassing level and recombine the gasses. The controlled overcharge is ok occasionally, as these are pure lead batteries and so almost no grid corrosion compared to doing this to a normal AGM. Then let it cool for 4 to 6 hours
Discharged a few Ah. Then charged at 14.70V and to 1000thC no problem. (68mA) termination. I actually took it to 55mA. Capacity now at least 80% as good as new.
So I charged again, at 14.70v till current stopped falling. CV for 12 hours. Then set it to 15.6V, and to 1A and left it for another 12 hours. Nothing to lose. Remember that this is 2 batts in parallel.
Obviously it was deeply sulfated. So slightly more volts, constant 1A current to desulfate it as far as possible. Because current is limited to .5A per battery, it can cope with the gassing level and recombine the gasses. The controlled overcharge is ok occasionally, as these are pure lead batteries and so almost no grid corrosion compared to doing this to a normal AGM. Then let it cool for 4 to 6 hours
Discharged a few Ah. Then charged at 14.70V and to 1000thC no problem. (68mA) termination. I actually took it to 55mA. Capacity now at least 80% as good as new.
-

Burgerman - Site Admin
- Posts: 65240
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Just dug an old chair out
Sat for a few hours. Quick top up in parallel, just to check. Dropped to 50mA per battery. You just need patience. Took several days to FULLY charge these.
-

Burgerman - Site Admin
- Posts: 65240
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Just dug an old chair out
"It didn't want to drop down to the 1000thC termination point"
You mentioned 1000thC termination point in other posts, though you have explained batteries very well. It was interesting with the paste etc plates
what is 1000thC?
When I did my NVQ2's in Electronics long time ago now, all I remember the tutor saying; was blah blah blah you only have to be able to build it, you don't need to understand it, blah blah blah
You get a bunch of NVQ2's which don't mean diddly in reality.
You mentioned 1000thC termination point in other posts, though you have explained batteries very well. It was interesting with the paste etc plates
what is 1000thC?
When I did my NVQ2's in Electronics long time ago now, all I remember the tutor saying; was blah blah blah you only have to be able to build it, you don't need to understand it, blah blah blah
You get a bunch of NVQ2's which don't mean diddly in reality.
-

Mechniki - Posts: 272
- Joined: 22 Oct 2013, 21:59
- Location: Dorking, Surrey
Re: Just dug an old chair out
C = capacity.
1000th of C on a 68Ah Odyssey such as I have here is 1000th of its capacity in Amps. So in this case thats a 68Ah or a 68,000mAh battery. So 1000th of this is 68mA.
So if you charge at ANY amp level, (more is better) then it will charge up and reach its recommended 14.40 to 14.70V CV stage voltage. So you just feed the battery say 14.7V until either 8 hours of CV has elapsed for safety. Or it reaches its termination current of 68mA. This is then 100% full, and no sulfates remain, and the battery needs no further low voltage float charge to get "full".
Most generic chargers do not have the capability of choosing such a accurate termination current. They have a one size fits non system. And stop charging and give you a green light at around 1.5 to .8 of an amp depending on who made the charger. At this point you are 97% to 99% charged. But that extra bit matters. Its leaves sulfates on the battery plate that have not been returned to the acid. So it then relies of float, to slowly complete the charge. But that really does take 16 hours or more. Which seldom happens. So the battery builds up a layer of lead sulfate on the plates, that turns to large non conducting crystals. And they are there for good. Not just on the surface, but deep inside those pasted plates.
Once a battery becomes sulfated by inadequate regular charging, or in my case an old abandoned chair, it has a very small capacity. And is high resistance. And the acid is weaker. Since the lead sulfate is on the plates and not returned by charging to the acid.
And the charge current NEVER falls to a really low level like 1000thC.
Charging at an elevated voltage causes gassing. And electrolyte loss, and can cause a battery to suffer thermal runaway. But it can return all the NON PERMANANT lead sulfate back to the acid. But it ruins the battery by distorting plates, loss of electrolyte, etc. So you can do it, but you must limit the current to a low level.
So I charged it as best I could normally. Then set a CC charge, with .5A per battery. The gasses produced are low enough at this current to be re-absorbed. And I set a 15.2 (have also used 15.6 on really knackered batteries) and do it for 16 hours, a day... Whatever it takes. It wont make a ruined battery well. It may make an old battery that has been used with a crap charger, better for a few months. And you will be able to tell as it will reach that low current on an 8 hour CV charge at the correct voltage. And your battery has an improved capacity for a while.
1000th of C on a 68Ah Odyssey such as I have here is 1000th of its capacity in Amps. So in this case thats a 68Ah or a 68,000mAh battery. So 1000th of this is 68mA.
So if you charge at ANY amp level, (more is better) then it will charge up and reach its recommended 14.40 to 14.70V CV stage voltage. So you just feed the battery say 14.7V until either 8 hours of CV has elapsed for safety. Or it reaches its termination current of 68mA. This is then 100% full, and no sulfates remain, and the battery needs no further low voltage float charge to get "full".
Most generic chargers do not have the capability of choosing such a accurate termination current. They have a one size fits non system. And stop charging and give you a green light at around 1.5 to .8 of an amp depending on who made the charger. At this point you are 97% to 99% charged. But that extra bit matters. Its leaves sulfates on the battery plate that have not been returned to the acid. So it then relies of float, to slowly complete the charge. But that really does take 16 hours or more. Which seldom happens. So the battery builds up a layer of lead sulfate on the plates, that turns to large non conducting crystals. And they are there for good. Not just on the surface, but deep inside those pasted plates.
Once a battery becomes sulfated by inadequate regular charging, or in my case an old abandoned chair, it has a very small capacity. And is high resistance. And the acid is weaker. Since the lead sulfate is on the plates and not returned by charging to the acid.
And the charge current NEVER falls to a really low level like 1000thC.
Charging at an elevated voltage causes gassing. And electrolyte loss, and can cause a battery to suffer thermal runaway. But it can return all the NON PERMANANT lead sulfate back to the acid. But it ruins the battery by distorting plates, loss of electrolyte, etc. So you can do it, but you must limit the current to a low level.
So I charged it as best I could normally. Then set a CC charge, with .5A per battery. The gasses produced are low enough at this current to be re-absorbed. And I set a 15.2 (have also used 15.6 on really knackered batteries) and do it for 16 hours, a day... Whatever it takes. It wont make a ruined battery well. It may make an old battery that has been used with a crap charger, better for a few months. And you will be able to tell as it will reach that low current on an 8 hour CV charge at the correct voltage. And your battery has an improved capacity for a while.
-

Burgerman - Site Admin
- Posts: 65240
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Just dug an old chair out
Charge current at 14.7V across 2x 68Ah odyssey batteries has now fallen even more to 78mA. Thats just 39mA per battery! Almost 2000thC. Those batts are 101% full...
It just takes time. This means its returned a little more of the sulfates caused by years of use and being dumped in the garage, back to the acid.
But its been at CV voltage for maybe 2 days almost on and off. This isn't healthy to do all the time, only to occasionally restore a flagging battery.
The normal limit for odyssey is 8 hours CV at 14.4 to 14.7V or till charge current falls to 1000th C whichever occurs first.
And voltages matter. Different types need accurate correct voltages. Likewise there are many float voltages.
-Overnight after a charge in DEEP CYCLE use where the charger stops CV too soon, or gel charging for slow charge use float cyclic use 13.7 or AGM 13.8.
-Overnight after a charge, safe for up to a week, 13.6V slower but safe for all.
-Longer than a week, up to a few years or standby, 13.3 to 13.4 depending on chemistry. Charge at CV 14.1/14.4 dep on chemistry every 4 months.
It just takes time. This means its returned a little more of the sulfates caused by years of use and being dumped in the garage, back to the acid.
But its been at CV voltage for maybe 2 days almost on and off. This isn't healthy to do all the time, only to occasionally restore a flagging battery.
The normal limit for odyssey is 8 hours CV at 14.4 to 14.7V or till charge current falls to 1000th C whichever occurs first.
And voltages matter. Different types need accurate correct voltages. Likewise there are many float voltages.
-Overnight after a charge in DEEP CYCLE use where the charger stops CV too soon, or gel charging for slow charge use float cyclic use 13.7 or AGM 13.8.
-Overnight after a charge, safe for up to a week, 13.6V slower but safe for all.
-Longer than a week, up to a few years or standby, 13.3 to 13.4 depending on chemistry. Charge at CV 14.1/14.4 dep on chemistry every 4 months.
-

Burgerman - Site Admin
- Posts: 65240
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Just dug an old chair out
I ended charge.
I choose MONITOR.
Its fallen as it should to 13.24V and will continue to do so. In the morning it will be 13.18 to 13.20.
In a week it will be 12.95 to 13.05 volts. If full, and healthy.
This is all BT by the way as the chair is the other side of a wall in the kitchen. Which now works 100%!
I choose MONITOR.
Its fallen as it should to 13.24V and will continue to do so. In the morning it will be 13.18 to 13.20.
In a week it will be 12.95 to 13.05 volts. If full, and healthy.
This is all BT by the way as the chair is the other side of a wall in the kitchen. Which now works 100%!
-

Burgerman - Site Admin
- Posts: 65240
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Just dug an old chair out
So 1000thC is Capacity. 1000th of an amp.
Assuming CC is constant charge
Assuming CV is constant Voltage - as it's battery related
and 'BT' is please?
Assuming CC is constant charge
Assuming CV is constant Voltage - as it's battery related
and 'BT' is please?
-

Mechniki - Posts: 272
- Joined: 22 Oct 2013, 21:59
- Location: Dorking, Surrey
Re: Just dug an old chair out
Bluetooth! I converted the charger, no wires.
-

Burgerman - Site Admin
- Posts: 65240
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Just dug an old chair out
Thank you!
-

Mechniki - Posts: 272
- Joined: 22 Oct 2013, 21:59
- Location: Dorking, Surrey
Re: Just dug an old chair out
>>So 1000thC is Capacity. 1000th of an amp.
1000thC is 1/000th of the battery capacity. So 70mA on a 70Ah (70,000mAh) battery. It is the termination current. End of charge point.
>>Assuming CC is constant charge
CC is Constant Current, the first stage of charge, defined by the power of the charger. An 8A charger charges at 8A till it reaches the CV voltage.
>>Assuming CV is constant Voltage - as it's battery related
Yes. The top continuous steady charge voltage, while waiting for current to fall to 1000thC...
>>and 'BT' is please?
Bluetooth.
In CYCLIC USE (us!) float is not required, if instead you continue CV (Absorption) stage until the Amps fall to a low 1000thC. As thats the fastest and best way to get a full charge in a limited period. But needs careful setup. And an 8 hour CV limit.
CV (or ABSORPTION if you are a dummy), Voltage depends on battery spec. But 14.1 or 14.4 or 14.7 for gel, AGM, and Odyssey.
The CV or ABSORPTION end point can be about 1A and followed by a l o n g float time. Or better, a low 1000thC point and no float needed.
CC is called BULK by dummies.
FLOAT is always called float, and is a lower voltage top up and long term maintenance CV.
1000thC is 1/000th of the battery capacity. So 70mA on a 70Ah (70,000mAh) battery. It is the termination current. End of charge point.
>>Assuming CC is constant charge
CC is Constant Current, the first stage of charge, defined by the power of the charger. An 8A charger charges at 8A till it reaches the CV voltage.
>>Assuming CV is constant Voltage - as it's battery related
Yes. The top continuous steady charge voltage, while waiting for current to fall to 1000thC...
>>and 'BT' is please?
Bluetooth.
In CYCLIC USE (us!) float is not required, if instead you continue CV (Absorption) stage until the Amps fall to a low 1000thC. As thats the fastest and best way to get a full charge in a limited period. But needs careful setup. And an 8 hour CV limit.
CV (or ABSORPTION if you are a dummy), Voltage depends on battery spec. But 14.1 or 14.4 or 14.7 for gel, AGM, and Odyssey.
The CV or ABSORPTION end point can be about 1A and followed by a l o n g float time. Or better, a low 1000thC point and no float needed.
CC is called BULK by dummies.
FLOAT is always called float, and is a lower voltage top up and long term maintenance CV.
-

Burgerman - Site Admin
- Posts: 65240
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Just dug an old chair out
Here is the real world version of that drawing/schematic above. Only the AMPS shown.
Still charging.
Charge at 30A. (the CC part) till it reaches CV level. Took 1 hour. With an 8A charger that would have taken 4 to 5 hours.
Then hold batt 14.7V and watch the amps naturally fall away as the battery charges up.
Its dropped from 30 to .8 of one amp. This is where your mobility charger stops CV and shows you a GREEN READY light. Usually before even this.
The battery is not yet fully charged, but you think it is. Its mobility charger will then either do nothing, or go to a lower float voltage that takes at least another 8 to 16 hours to achieve a full charge if ever.
But my PL8 is configured to keep going till it sees a low 100mA. 2x parallel Odyssey batts. Less current than even 1000thC. And then to charge at at CC 100mA for 1 hour on top. It will then be FULLY charged.
Still charging.
Charge at 30A. (the CC part) till it reaches CV level. Took 1 hour. With an 8A charger that would have taken 4 to 5 hours.
Then hold batt 14.7V and watch the amps naturally fall away as the battery charges up.
Its dropped from 30 to .8 of one amp. This is where your mobility charger stops CV and shows you a GREEN READY light. Usually before even this.
The battery is not yet fully charged, but you think it is. Its mobility charger will then either do nothing, or go to a lower float voltage that takes at least another 8 to 16 hours to achieve a full charge if ever.
But my PL8 is configured to keep going till it sees a low 100mA. 2x parallel Odyssey batts. Less current than even 1000thC. And then to charge at at CC 100mA for 1 hour on top. It will then be FULLY charged.
-

Burgerman - Site Admin
- Posts: 65240
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Just dug an old chair out
And a few hours later, finally reached 98mA. And stopped CV.
-

Burgerman - Site Admin
- Posts: 65240
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Just dug an old chair out
98mA for one pair together is a very small current .
They will hold a charge .
They will hold a charge .
- shirley_hkg
- Posts: 3944
- Joined: 31 Dec 2010, 13:42
Re: Just dug an old chair out
Yep. Dropped to 70 after another hour or two.
Heres the thing.
If your battery DOES NOT drop this low, it is sulfated. Much of that isn't permanent. A slow controlled overcharge at CC can fix it.
To fix it, and gain a lot of performance and charge efficiency, do a one off 2 day controlled low current 15.2V max overcharge at .5A AFTER charging normally. Even on old gels. You have nothing to lose. If current falls to almost zero, stop. Remember that at higher voltages like this current often remains high. So now and again, give it 10 mins rest, and try charging at the correct CV voltage. See what current it drops to.
The Shirley power supply NOT set to 3 stage, can do that. Any adjustable supply can.
What you are then doing, is removing the deeply embedded sulfates from the lead pasted grids, whilst not damaging the battery (much). The idea is that at .5A max the higher voltage doesn't matter because the recombinant chemistry can cope with the gas produced at this low current level. And the controlled continual slight CC overcharge forces any non permanent sulfates to return to the acid. Dangers are loss of electrolyte and grid corrosion. But if the battery is dying from sulfation due to undercharge then thats a bigger problem.
Heres the thing.
If your battery DOES NOT drop this low, it is sulfated. Much of that isn't permanent. A slow controlled overcharge at CC can fix it.
To fix it, and gain a lot of performance and charge efficiency, do a one off 2 day controlled low current 15.2V max overcharge at .5A AFTER charging normally. Even on old gels. You have nothing to lose. If current falls to almost zero, stop. Remember that at higher voltages like this current often remains high. So now and again, give it 10 mins rest, and try charging at the correct CV voltage. See what current it drops to.
The Shirley power supply NOT set to 3 stage, can do that. Any adjustable supply can.
What you are then doing, is removing the deeply embedded sulfates from the lead pasted grids, whilst not damaging the battery (much). The idea is that at .5A max the higher voltage doesn't matter because the recombinant chemistry can cope with the gas produced at this low current level. And the controlled continual slight CC overcharge forces any non permanent sulfates to return to the acid. Dangers are loss of electrolyte and grid corrosion. But if the battery is dying from sulfation due to undercharge then thats a bigger problem.
-

Burgerman - Site Admin
- Posts: 65240
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Just dug an old chair out
Took out 33Ah yesterday.
Today putting it back, and choose 36A and 14.7V.
After just over an hour, much of it is back. And we are down to 7.5A charge... The same as a stock uselessly slow mobility charger at max power!
Remember that we do this overnight, so about 8 hours. With a fast charger at 30 to 40A the bulk of the Ah is back in in an hour. Leaving 7 hours to let the ever slowing CV stage to get as much back in before you unplug the charger. With a 5 or 8A mobility charger you waste 5 hours at CC going slow, before you ever reach the CV part. And so run out of time to complete the charge. But it gives you a green regardless! Sulfation city ahead.
Here is Amps V time. Down to the Amp point where a mobility charger begins...
So after 1 hour you have a huge head start of 29Ah. With 5Ah remaining. If I charged at 7.5Amps like a mobility charger it would have taken 5 hours to get to this point. Leaving just 3 for the CV stage before you get up.
This is why, ANDERSONS!
And a powerful charger. It allows 7 hours of CV instead, so gives a much more complete charge over an 8 hour night.
Today putting it back, and choose 36A and 14.7V.
After just over an hour, much of it is back. And we are down to 7.5A charge... The same as a stock uselessly slow mobility charger at max power!
Remember that we do this overnight, so about 8 hours. With a fast charger at 30 to 40A the bulk of the Ah is back in in an hour. Leaving 7 hours to let the ever slowing CV stage to get as much back in before you unplug the charger. With a 5 or 8A mobility charger you waste 5 hours at CC going slow, before you ever reach the CV part. And so run out of time to complete the charge. But it gives you a green regardless! Sulfation city ahead.
Here is Amps V time. Down to the Amp point where a mobility charger begins...
So after 1 hour you have a huge head start of 29Ah. With 5Ah remaining. If I charged at 7.5Amps like a mobility charger it would have taken 5 hours to get to this point. Leaving just 3 for the CV stage before you get up.
This is why, ANDERSONS!
And a powerful charger. It allows 7 hours of CV instead, so gives a much more complete charge over an 8 hour night.
-

Burgerman - Site Admin
- Posts: 65240
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Just dug an old chair out
FINAL step before storing the old chair back in the garage.
MEASURE AH OUT!!!
We need to know the condition of these batteries after approx 250 cycles, and 10 year calendar life, inc being uncharged for a year in my garage. Before being abandoned in favour of lithium, etc they did around 200 to 250 cycles. And are now a decade old. They WERE stored for 9 of those years on a low 13.4 float.
So lets see whats left of them.
They are currently on a discharge of 7.75A. As a pair, in parallel. We are then discharging at around a 17 hour rate.
So thats 2x Odyssey PC1500, which should give: 68Ah@20Hrate, or 65Ah@10H rate. To 10.2V.
So would I expect to get about 66Ah from this set if they were as good as new. right now we are up to 99Ah or about 50Ah per battery. Ans still going.
MEASURE AH OUT!!!
We need to know the condition of these batteries after approx 250 cycles, and 10 year calendar life, inc being uncharged for a year in my garage. Before being abandoned in favour of lithium, etc they did around 200 to 250 cycles. And are now a decade old. They WERE stored for 9 of those years on a low 13.4 float.
So lets see whats left of them.
They are currently on a discharge of 7.75A. As a pair, in parallel. We are then discharging at around a 17 hour rate.
So thats 2x Odyssey PC1500, which should give: 68Ah@20Hrate, or 65Ah@10H rate. To 10.2V.
So would I expect to get about 66Ah from this set if they were as good as new. right now we are up to 99Ah or about 50Ah per battery. Ans still going.
-

Burgerman - Site Admin
- Posts: 65240
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Just dug an old chair out
10.2V sounds like draining to empty.
What voltage does a chair controller usually decide it's flat and start flashing red lights?
What voltage does a chair controller usually decide it's flat and start flashing red lights?
-

foghornleghorn - Posts: 638
- Joined: 20 Mar 2018, 16:29
- Location: South East England
Re: Just dug an old chair out
Empty is a matter of how discharged you decide empty is. And that voltage depends on discharge rate too. Discharge in 2 hours and 9V is not empty. Discharge in 24 hours and 10.8V is empty. When does a chair stop? They vary, and average voltage depression, but at about 11.5V average unloaded it dies, 10V peak under load.
This battery has 2 capacity ratings.
10h rate is 65Ah.
20h rate is 68Ah.
THIS discharge was at the 17 hour rate. Actually took 16...
Results are for 2 parallel batteries so divide by 2:
10.8v = 117.1Ah
10.5v = 118.9Ah
10.2v = 120.0Ah RATED
10.0v = 121.0Ah
Actual rated storage capacity at 15 hour rate as new would be 67Ah. I just measured 60Ah, so this battery is missing 7Ah. About an 11% capacity loss over its life. The PL8 is a very useful tool, IF you know how and what you are measuring.
This battery has 2 capacity ratings.
10h rate is 65Ah.
20h rate is 68Ah.
THIS discharge was at the 17 hour rate. Actually took 16...
Results are for 2 parallel batteries so divide by 2:
10.8v = 117.1Ah
10.5v = 118.9Ah
10.2v = 120.0Ah RATED
10.0v = 121.0Ah
Actual rated storage capacity at 15 hour rate as new would be 67Ah. I just measured 60Ah, so this battery is missing 7Ah. About an 11% capacity loss over its life. The PL8 is a very useful tool, IF you know how and what you are measuring.
-

Burgerman - Site Admin
- Posts: 65240
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Just dug an old chair out
Peukert means less Ah as you discharge faster.
But what screws up a powerchair is the voltage drop under high current conditions. ON TOP of the reduced Ah
Remember that a 5Ah battery for e.g., can have 200A pulled from it as you turn or climb a ramp. Thats 4C. Look at the voltages on the graph. We take power in pulses.
So the voltage drop, and Ah reduction of a grp22 over a grp24 battery makes a simply HUGE combined performance reduction. Out of all proportion if you simply look at battery Ah. And this also rapidly deteriorates the smaller battery as well, accelerating the decline.
Remember that at 12V you get red flashing lights on most chairs.
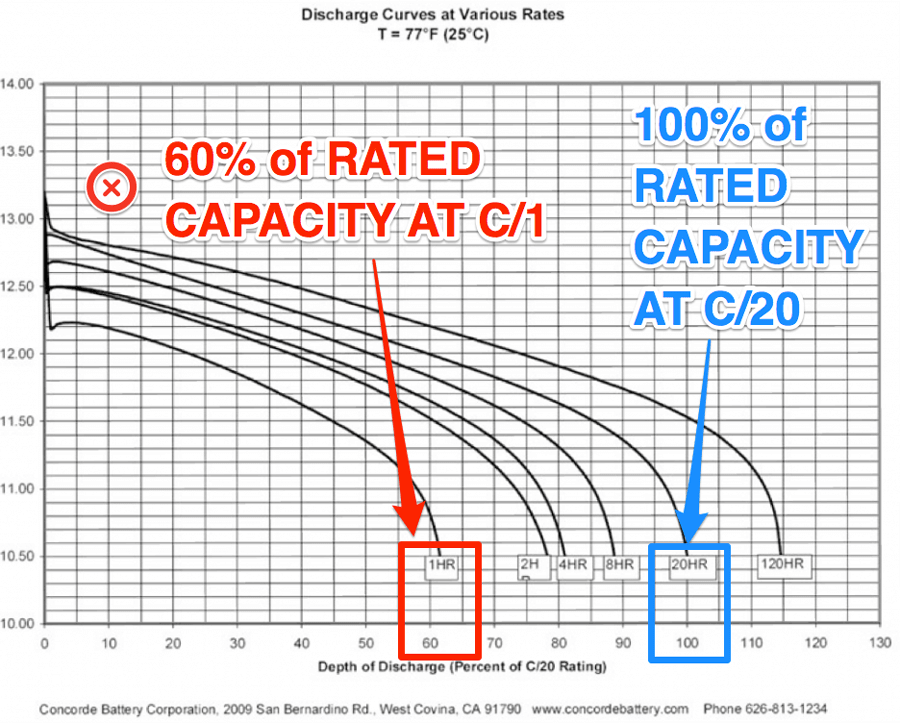
But what screws up a powerchair is the voltage drop under high current conditions. ON TOP of the reduced Ah
Remember that a 5Ah battery for e.g., can have 200A pulled from it as you turn or climb a ramp. Thats 4C. Look at the voltages on the graph. We take power in pulses.
So the voltage drop, and Ah reduction of a grp22 over a grp24 battery makes a simply HUGE combined performance reduction. Out of all proportion if you simply look at battery Ah. And this also rapidly deteriorates the smaller battery as well, accelerating the decline.
Remember that at 12V you get red flashing lights on most chairs.

-

Burgerman - Site Admin
- Posts: 65240
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Just dug an old chair out
Found this now
So chair starts to panic when batteries are about 11.7v
I've pickled my brain trying to work out how much will have actually been used for example in 4h to get to 11.7v, if the capacity test runs down to 10.2v at the same rate as you will be halfway along a graph
Pilot+ Technical Manual
The battery gauge typically starts to flash slowly when the battery voltage falls below 23.3V whilst the
wheelchair is driving on a level surface.
So chair starts to panic when batteries are about 11.7v
I've pickled my brain trying to work out how much will have actually been used for example in 4h to get to 11.7v, if the capacity test runs down to 10.2v at the same rate as you will be halfway along a graph
-

foghornleghorn - Posts: 638
- Joined: 20 Mar 2018, 16:29
- Location: South East England
53 posts
• Page 1 of 2 • 1, 2
Return to Everything Powerchair
Who is online
Users browsing this forum: No registered users and 26 guests