Battgo Cell Charge Checker.
25 posts
• Page 1 of 1
Battgo Cell Charge Checker.
I got one of these to check my 30Ah expresso build pack but can't get it to work.
The problem is the connector from the pack has the wrong polarity for the Battgo? I used another lead, cut and swapped the + and - wires changing the polarity from the pack but that did not work.
The monitor seems to work OK, I have powered it up using its main input and also powered it up from the pins just by providing power to the negative pin and then the live to any other pin.
https://www.banggood.com/ISDT-BattGo-BG ... rehouse=CN
The problem is the connector from the pack has the wrong polarity for the Battgo? I used another lead, cut and swapped the + and - wires changing the polarity from the pack but that did not work.
The monitor seems to work OK, I have powered it up using its main input and also powered it up from the pins just by providing power to the negative pin and then the live to any other pin.
https://www.banggood.com/ISDT-BattGo-BG ... rehouse=CN
I'm a dude playing a dude disguised as another dude.
- Dan
- Posts: 544
- Joined: 10 Nov 2011, 03:03
- Location: Northern Ireland
Re: Battgo Cell Charge Checker.
You need to reverse all node wires not just +ve and -ve.
-

Scooterman - Posts: 2773
- Joined: 03 Jul 2016, 10:11
- Location: Camberley England UK
Re: Battgo Cell Charge Checker.
I thought that might be the case.
Thanks.
Thanks.
I'm a dude playing a dude disguised as another dude.
- Dan
- Posts: 544
- Joined: 10 Nov 2011, 03:03
- Location: Northern Ireland
Re: Battgo Cell Charge Checker.
Turn plug 180...
-

Burgerman - Site Admin
- Posts: 65305
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Battgo Cell Charge Checker.
Tried turning it around but the little clip on the plug will not let it push in also the pins look like they are offset a bit. Anyway I should cut the latch off the plug and give it a go.
I'm a dude playing a dude disguised as another dude.
- Dan
- Posts: 544
- Joined: 10 Nov 2011, 03:03
- Location: Northern Ireland
Re: Battgo Cell Charge Checker.
It is hard to tell what kind of connector it uses from the pictures on the web site that you posted.
The obvious picture shows a 6s cable plugged into the meter, yet the description says that it support 8s.
From the picture, which is far from definitive, I would guess it might use a JST-XH connector. These are common in the RC hobby domain.
If the device accepts a 9 pin JST-XH connector with female pins, it should be fairly easy to remove the cable's pins from the plastic housing and reinsert them in the desired order. A quick search on youtube will show you examples of how to remove JST-XH female pins from the plastic housing and reinsert them.
I use this cellmeter, which allows me to look at the voltage for each individual cell in an 8s battery pack. My cellmeter has a male JST-XH connector which accepts a female JST-XH connector. It seems to work pretty well, showing a voltage within about 30 millivolts of what my PL8 shows.
https://www.amazon.com/gp/product/B073W ... UTF8&psc=1
Steve
The obvious picture shows a 6s cable plugged into the meter, yet the description says that it support 8s.
From the picture, which is far from definitive, I would guess it might use a JST-XH connector. These are common in the RC hobby domain.
If the device accepts a 9 pin JST-XH connector with female pins, it should be fairly easy to remove the cable's pins from the plastic housing and reinsert them in the desired order. A quick search on youtube will show you examples of how to remove JST-XH female pins from the plastic housing and reinsert them.
I use this cellmeter, which allows me to look at the voltage for each individual cell in an 8s battery pack. My cellmeter has a male JST-XH connector which accepts a female JST-XH connector. It seems to work pretty well, showing a voltage within about 30 millivolts of what my PL8 shows.
https://www.amazon.com/gp/product/B073W ... UTF8&psc=1
Steve
Permobil F5 Corpus 3G
Permobil C500s VS
Permobil C500 Corpus 3G
Permobil C350 Corpus 3G
Magic Mobility X4 with 176 Ah LiFePO4
Permobil C500s VS
Permobil C500 Corpus 3G
Permobil C350 Corpus 3G
Magic Mobility X4 with 176 Ah LiFePO4
- swalker
- Posts: 550
- Joined: 23 Jul 2018, 22:57
- Location: Vail, Colorado, USA
Re: Battgo Cell Charge Checker.
It still wont tell you whats left. Different brands of cells very by more than 100mV across the range. And if they didnt, it still wont help as temperature alone means it will read at 50% full when its actually 90 or 30% since the discharge curve is basically flat. It varies more by manufacturer, load, and temperature than by state of charge. In fact as you use the battery its voltage may increase as it warms up...
What it is useful for is as an alarm at the point where it drops fast below around 3.1V per cell. So set it to around 2.9V depending on internal resistance, C rate, temperature, load current this might need increasing or reducing to 2.5V at the lowest.
What it is useful for is as an alarm at the point where it drops fast below around 3.1V per cell. So set it to around 2.9V depending on internal resistance, C rate, temperature, load current this might need increasing or reducing to 2.5V at the lowest.
-

Burgerman - Site Admin
- Posts: 65305
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Battgo Cell Charge Checker.
Well it turns out I'm an idiot and not matter what way the plug was twisted it did not fit! The pins are to close together.
Swalker you were right. I think I need glasses
youtu.be/UIYCKYfNkiE
Its the end of the revolectrix charge cable that plugs into the PL8.
Swalker you were right. I think I need glasses
youtu.be/UIYCKYfNkiE
Its the end of the revolectrix charge cable that plugs into the PL8.
- Attachments
-
- 29.jpg (34.16 KiB) Viewed 5212 times
I'm a dude playing a dude disguised as another dude.
- Dan
- Posts: 544
- Joined: 10 Nov 2011, 03:03
- Location: Northern Ireland
-

steves1977uk - Posts: 4342
- Joined: 20 Oct 2011, 21:47
- Location: Wells next the Sea, Norfolk, UK
Re: Battgo Cell Charge Checker.
Thanks. I think I'm sorted now.
I'm a dude playing a dude disguised as another dude.
- Dan
- Posts: 544
- Joined: 10 Nov 2011, 03:03
- Location: Northern Ireland
Re: Battgo Cell Charge Checker.
I don't think so , Steve. It's not just about sizes.
Totally different connectors.
Totally different connectors.
- shirley_hkg
- Posts: 3949
- Joined: 31 Dec 2010, 13:42
Re: Battgo Cell Charge Checker.
@Shirley, I agree it isn't just about the type of shell connector, but the pin-to-pin pitch distance too. 
Steve
Steve
-

steves1977uk - Posts: 4342
- Joined: 20 Oct 2011, 21:47
- Location: Wells next the Sea, Norfolk, UK
Re: Battgo Cell Charge Checker.
Hi,
This one from Aliexpress is perfect:
8S JST-XH Connector Balance Silicone wire Cable 100cm 9PIN 1Meter 20in 20#AWG.
US $4.95
Furio.
This one from Aliexpress is perfect:

8S JST-XH Connector Balance Silicone wire Cable 100cm 9PIN 1Meter 20in 20#AWG.
US $4.95
Furio.
- Furio
- Posts: 184
- Joined: 24 Dec 2011, 18:10
- Location: India
Re: Battgo Cell Charge Checker.
Does a battery checker know what type cell chemistry it's monitoring, does it make a difference? Has anyone else got one of these types? I know Steve has. If so why is mine lousy accuracy? 7 cells show various voltages in the same ballpark, but one much lower than the rest. And yet the PL8 and Fluke show them all the same except one cell that always sits a couple of millivolts higher than the rest.
Surely it's not rocket science for the cell checker to check voltages accurately? I accept that it's only cheap and voltage may not match a multimeter, but they should surely all show the same, say within 2-3mV?
Useless piece of junk



Surely it's not rocket science for the cell checker to check voltages accurately? I accept that it's only cheap and voltage may not match a multimeter, but they should surely all show the same, say within 2-3mV?
Useless piece of junk




-

Scooterman - Posts: 2773
- Joined: 03 Jul 2016, 10:11
- Location: Camberley England UK
Re: Battgo Cell Charge Checker.
Even your multimeter cant reliably measure to 1mV unless its very well and recently calibrated and high quality. Even the different metals in a test lead connector can generate a couple of mV.
I have one of these... https://www.fluke.com/en-gb/product/ele ... /fluke-289
And with a recent calibration certificate. It gets close. But it needs to be on, in a temp stable room for half an hour before its accurate to 0.025% of its scale +/- a few digits on the end figure. And it costs the same as 2 PL8s and then some.
Those cheap cell logs, are like buying 8 voltmeters for a manufacturing cost of a few pence. Theres a BIG difference between resolution, and accuracy.
You only need them to show a drop at the end from 3.2xV down to say 3V so as long as you are only using them as an alarm to stop battery damage theres no problem The danger is that people insist on thinking that they can monitor a battery like a fuel gauge based on volts. You cant.
If you wanted to try, then you would need to do these things.
MAP the voltages, under a 1C discharge, and a 2, 3C discharge under discharge conditions with a new battery, and at various temperatures. Then do the same in stages with zero current, breaks so as to have an unloaded voltage graph on discharge. So about 20 seperate discharge curves and graphs.
Then...
Fit a temp sensor to your battery. And use a computer to monitor current, and voltage, and temperature. And with a little averaging, and a bit of maths, you could have a fuel guage based on the stored maps. Thats the ONLY way to do this. And that will notwork for lead. And will be completely different on every different type of lithium ion phosphate battery depending on the exact chemistry and percentages of each chemicals, types of anode/cathode etc used. So it will be individual to YOUR pack.
I have one of these... https://www.fluke.com/en-gb/product/ele ... /fluke-289
And with a recent calibration certificate. It gets close. But it needs to be on, in a temp stable room for half an hour before its accurate to 0.025% of its scale +/- a few digits on the end figure. And it costs the same as 2 PL8s and then some.
Those cheap cell logs, are like buying 8 voltmeters for a manufacturing cost of a few pence. Theres a BIG difference between resolution, and accuracy.
You only need them to show a drop at the end from 3.2xV down to say 3V so as long as you are only using them as an alarm to stop battery damage theres no problem The danger is that people insist on thinking that they can monitor a battery like a fuel gauge based on volts. You cant.
If you wanted to try, then you would need to do these things.
MAP the voltages, under a 1C discharge, and a 2, 3C discharge under discharge conditions with a new battery, and at various temperatures. Then do the same in stages with zero current, breaks so as to have an unloaded voltage graph on discharge. So about 20 seperate discharge curves and graphs.
Then...
Fit a temp sensor to your battery. And use a computer to monitor current, and voltage, and temperature. And with a little averaging, and a bit of maths, you could have a fuel guage based on the stored maps. Thats the ONLY way to do this. And that will notwork for lead. And will be completely different on every different type of lithium ion phosphate battery depending on the exact chemistry and percentages of each chemicals, types of anode/cathode etc used. So it will be individual to YOUR pack.
-

Burgerman - Site Admin
- Posts: 65305
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Battgo Cell Charge Checker.
Does a battery checker know what type cell chemistry it's monitoring, does it make a difference?
None can ever know automatically. Some can be set to lifepo4, lipo, or lithium ion. All three umbrella terms vastly different. And even 3 different makes of "lifepo4" or life are different enough from each other that you cannot use these as a fuel gauge even if they have the same charge voltages...
For e.g. the lithium ion batteries in the tesla, are more graphite/nickel than lico style laptop batteries... But both are "lithium ion". Theres thousands of variations of lithiums. But 3 main umbrella terms.
-

Burgerman - Site Admin
- Posts: 65305
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Battgo Cell Charge Checker.
You also might read this.
Accuracy refers to the largest allowable error that occurs under specific operating conditions.
Accuracy is expressed as a percentage and indicates how close the displayed measurement is to the actual (standard) value of the signal measured. Accuracy requires a comparison to an accepted industry standard.
The accuracy of a specific digital multimeter is more or less important depending on the application. For example, most AC power line voltages vary ±5% or more. An example of this variation is a voltage measurement taken at a standard 115 V AC receptacle. If a digital multimeter is only used to check if a receptacle is energized, a DMM with a ±3% measurement accuracy is appropriate.
Some applications, such as calibration of automotive, medical aviation or specialized industrial equipment, may require higher accuracy. A reading of 100.0 V on a DMM with an accuracy of ±2% can range from 98.0 V to 102.0 V. This may be fine for some applications, but unacceptable for sensitive electronic equipment.
Accuracy may also include a specified amount of digits (counts) added to the basic accuracy rating. For example, an accuracy of ±(2%+2) means that a reading of 100.0 V on the multimeter can be from 97.8 V to 102.2 V. Use of a DMM with higher accuracy allows a great number of applications.
Basic dc accuracy of Fluke handheld digital multimeters ranges from 0.5% to 0.025%.
And understand this:
Basic dc accuracy of Fluke handheld digital multimeters ranges from 0.5% to 0.025%.
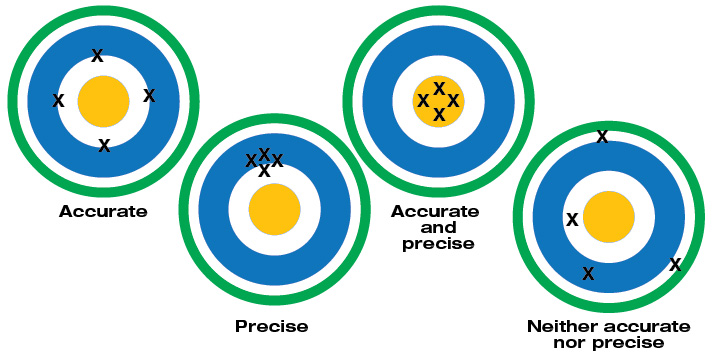
Accuracy refers to the largest allowable error that occurs under specific operating conditions.
Accuracy is expressed as a percentage and indicates how close the displayed measurement is to the actual (standard) value of the signal measured. Accuracy requires a comparison to an accepted industry standard.
The accuracy of a specific digital multimeter is more or less important depending on the application. For example, most AC power line voltages vary ±5% or more. An example of this variation is a voltage measurement taken at a standard 115 V AC receptacle. If a digital multimeter is only used to check if a receptacle is energized, a DMM with a ±3% measurement accuracy is appropriate.
Some applications, such as calibration of automotive, medical aviation or specialized industrial equipment, may require higher accuracy. A reading of 100.0 V on a DMM with an accuracy of ±2% can range from 98.0 V to 102.0 V. This may be fine for some applications, but unacceptable for sensitive electronic equipment.
Accuracy may also include a specified amount of digits (counts) added to the basic accuracy rating. For example, an accuracy of ±(2%+2) means that a reading of 100.0 V on the multimeter can be from 97.8 V to 102.2 V. Use of a DMM with higher accuracy allows a great number of applications.
Basic dc accuracy of Fluke handheld digital multimeters ranges from 0.5% to 0.025%.
And understand this:
Basic dc accuracy of Fluke handheld digital multimeters ranges from 0.5% to 0.025%.

-

Burgerman - Site Admin
- Posts: 65305
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Battgo Cell Charge Checker.
I used to advocate installing a cell log, but never have a chance to it , since 2012.
With enough Ah installed , I've never been able to run it low enough , except skip re-charging deliberately.
I'm comfortable with a simple voltmetre.
With enough Ah installed , I've never been able to run it low enough , except skip re-charging deliberately.
I'm comfortable with a simple voltmetre.
- shirley_hkg
- Posts: 3949
- Joined: 31 Dec 2010, 13:42
Re: Battgo Cell Charge Checker.
For what its worth...
This is a 5S lipo.
The total voltage reads 19.18 on the battgo. Which is quite close to the true 19.122 figure.
Within 58mV of the correct figure.
Cell voltages varied up to 10mV +/minus, usually low. compared to the true figure. A spread of 20mV!
When I try this on my PL8s, they are all within around 3mV.
nOTE VERY SHARP NEEDLE ADAPTERS... tHROUGH CABLE INSULATION, OR INTO CELL CONNECTORS NO PROB. Ooops, caps lock...
Image is after it has been balancing for 6 hours. Now its unbalanced but reads as if it is...
This is a 5S lipo.
The total voltage reads 19.18 on the battgo. Which is quite close to the true 19.122 figure.
Within 58mV of the correct figure.
Cell voltages varied up to 10mV +/minus, usually low. compared to the true figure. A spread of 20mV!
When I try this on my PL8s, they are all within around 3mV.
nOTE VERY SHARP NEEDLE ADAPTERS... tHROUGH CABLE INSULATION, OR INTO CELL CONNECTORS NO PROB. Ooops, caps lock...
Image is after it has been balancing for 6 hours. Now its unbalanced but reads as if it is...
-

Burgerman - Site Admin
- Posts: 65305
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Battgo Cell Charge Checker.
What do you think of this? It's small and dinky might double as a voltage checker and phone charger?

https://www.ebay.co.uk/itm/DC4-30V-USB- ... QCXFk5mbBA
I was think...
Get a short USB lead extension lead. Cut the male end off and wire to batteries. Then plug voltmeter into female USB on flying lead. It'd then double as a voltage checker and phone charger and is nice and dinky.
Yes/No?

https://www.ebay.co.uk/itm/DC4-30V-USB- ... QCXFk5mbBA
I was think...
Get a short USB lead extension lead. Cut the male end off and wire to batteries. Then plug voltmeter into female USB on flying lead. It'd then double as a voltage checker and phone charger and is nice and dinky.
Yes/No?
-

Scooterman - Posts: 2773
- Joined: 03 Jul 2016, 10:11
- Location: Camberley England UK
Re: Battgo Cell Charge Checker.
Dunno what its measuring, USB is supposed to be 5.00V. Or how accurate that is.
-

Burgerman - Site Admin
- Posts: 65305
- Joined: 27 May 2008, 21:24
- Location: United Kingdom
Re: Battgo Cell Charge Checker.
Burgerman wrote:Dunno what its measuring, USB is supposed to be 5.00V. Or how accurate that is.
I haven't the foggiest either!
I thought on hindsight it might blow up my phone by pumping 24-26v into it

-

Scooterman - Posts: 2773
- Joined: 03 Jul 2016, 10:11
- Location: Camberley England UK
Re: Battgo Cell Charge Checker.
Those USB volts/amps checkers are for phones/Nintendo DS and the likes to check if the device is drawing any power when doing repairs on them. MyMateVince on Youtube uses one.
youtu.be/Akz2CaEuP2A
Steve
youtu.be/Akz2CaEuP2A
Steve
-

steves1977uk - Posts: 4342
- Joined: 20 Oct 2011, 21:47
- Location: Wells next the Sea, Norfolk, UK
Re: Battgo Cell Charge Checker.
Scooterman wrote:What do you think of this? It's small and dinky might double as a voltage checker and phone charger?
https://www.ebay.co.uk/itm/DC4-30V-USB- ... QCXFk5mbBA
I was think...
Get a short USB lead extension lead. Cut the male end off and wire to batteries. Then plug voltmeter into female USB on flying lead. It'd then double as a voltage checker and phone charger and is nice and dinky.
Yes/No?
All but certainly NO.... It isn't intended to do any voltage conversion - just tell you if the USB connector you are plugging it into is supplying the correct voltage, and possibly current as called for in the USB specs.... It doesn't say what the input voltage expectations are, but usually things like that are designed with the idea in mind that the input is "reasonably close" to what it would expect for a 'good' reading, or that it's LESS (and will give a bad reading)
My expectation if you connected it to a 24V chair battery would involve significant amounts of smoke, if not flames.... At best it would either show no reading or BAD....
ex-Gooserider
T-5, ASIA-B
Jazzy 1100
Jazzy Select 6
Quickie Q-7
Invacare Mariner
Want to make / get a better chair, ideally one that stands.
Jazzy 1100
Jazzy Select 6
Quickie Q-7
Invacare Mariner
Want to make / get a better chair, ideally one that stands.
-

ex-Gooserider - Posts: 5969
- Joined: 15 Feb 2011, 06:17
- Location: Billerica, MA. USA
25 posts
• Page 1 of 1
Return to Everything Powerchair
Who is online
Users browsing this forum: shirley_hkg and 20 guests